If using fluorescent detection, be sure to remove Ponceau S before immunostaining as this can autofluoresce. Wash extensively in buffer between all steps. Residual unbound antibodies or other reagents remaining between steps can produce a high background. Keep on ice throughout the western blot process. We recommend blocking 3–5% non-fat dry milk, BSA, or normal serum for 1 hr at room temperature. Increase the blocking incubation period and consider changing the blocking agent. If using phospho-specific antibodies, block with BSA instead of milk.īlocking of non-specific binding may be insufficient. Note that phospho-specific antibodies may react with a milk blocking agent due to the presence of the phosphoprotein casein. The secondary antibody may be binding to the blocking reagent.Īdd a mild detergent such as Tween 20 to the incubation and washing buffer. Primary antibody concentration may be too high.ĭilute the antibody further to its optimal concentration. Try a secondary antibody that has been pre-adsorbed against the lg of the species of your samples. Make sure you use a secondary antibody raised in a different species to your sample. Run a control without any primary antibody. The secondary antibody may be binding non-specifically For example, sodium azide is an inhibitor of HRP, so it is unsuitable for use with HRP-conjugated antibodies.Ĭheck your buffers don't contain any incompatible reagents, and change the buffer if needed. Some buffers contain reagents that may interfere with detection. 
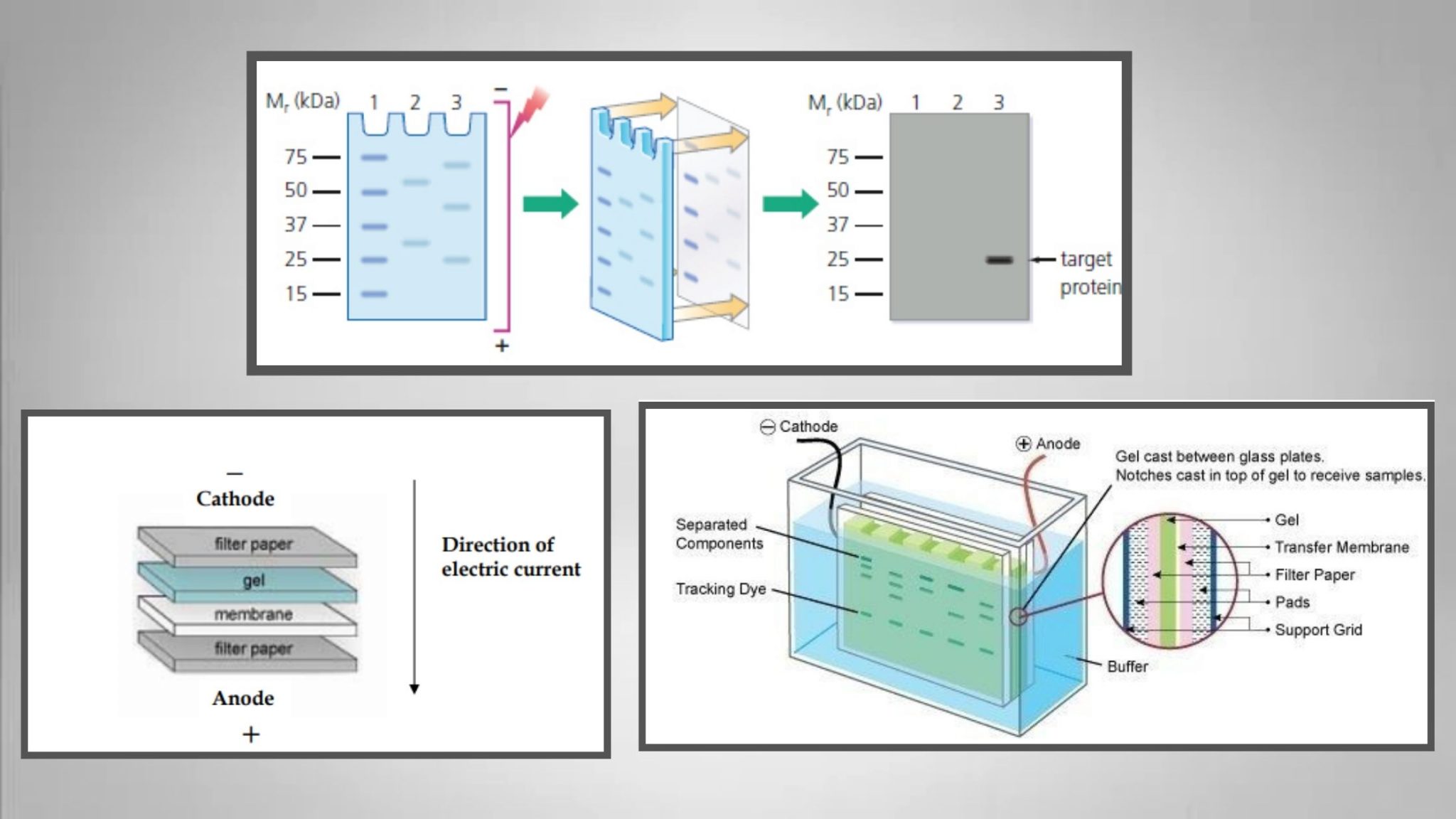
Make sure you use fresh primary and secondary antibodies for each experiment the effective antibody concentration is lowered after each use.īuffers may be incompatible with the detection method. Overuse of antibodies has reduced their effectiveness. Use an enrichment step to maximize the signal (eg prepare nuclear lysates for a nuclear protein). Make sure you load at least 20–30 µg protein per lane, use protease inhibitors, and run the recommended positive control. Incubate the sample for longer with the antibody (eg overnight) at 4☌.Ĭheck the scientific literature to see if the protein is expected in your cell line. Not enough antibody is bound to the protein.Īdd a higher concentration of primary antibody Make sure that the isotypes of the primary and secondary are compatible. Make sure you use a secondary antibody raised against the primary antibody species. The primary antibody and the secondary antibody are not compatible. If only the sample lanes are difficult to see, and the molecular weight ladder is unaffected, this suggests there are issues detecting the protein of interest. This may require some optimization to get right.īands in the sample lanes are faint or have no signal Try imaging the blot again with a longer exposure time. There may not be enough exposure time when imaging the blot. You may have used the wrong filter settings for detection.Įnsure you set the instrument to read the correct wavelengths. Store and handle fluorophores and fluorophore-conjugated antibodies in the dark and minimize light exposure by wrapping the vial in foil. If using fluorescent detection, the fluorophore may have been damaged by too much light exposure. Reagents may have lost activity due to improper storage and handling.Ĭheck the storage instructions for your products on the datasheet. Use fresh, sterile buffer (eg our sterile PBS). The wash or incubation buffer is contaminated with bacteria. Reduce the duration or number of washing steps. Washing with buffer between steps is necessary, but sometimes washing too aggressively can remove detection reagents. If using a PVDF membrane, make sure you pre-soak the membrane in methanol and then in transfer buffer. If the proteins have not transferred effectively, check the transfer was performed in the right direction ( see diagram). Problems with transfer of proteins to the membrane.Ĭheck the transfer was successful using a reversible stain such as Ponceau S before immunostaining. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
